Pharmacogenomics: A tool of choice to treat ADHD
When a child is diagnosed with attention deficit disorder (ADD) or attention deficit hyperactivity disorder (ADHD), it is possible to relieve the symptoms. One solution is to prescribe a pharmacological treatment to complement other strategies. The problem lies in finding the right molecule at the right dosage for the treatment to be fully effective. Rather than relying solely on a traditional trial-and-error approach, pharmacogenomics provides information that can assist in choosing a medication.
ADHD: A combined treatment approach
To achieve better results when treating children with ADHD, a combined approach is often recommended. It is often based on a combination of strategies:
- Putting behavioural strategies in place
- Integrating the child into an appropriate school environment
- Establishing routines and healthy habits (diet, sleep, physical activity and screen time)
- Taking medication [1]
The risks of taking medication
The choice of whether or not to give a child medication is a very personal and difficult decision for parents. In addition, among the many medications that can help children living with ADHD, it is not easy to determine which one will work best for them.
The trial-and-error process
Finding the right molecule, the right dosage or the best combination of treatments is sometimes a lengthy process. While some children feel the medication’s benefits on the first try, others may have to be patient and go through a long series of trials and errors.
In addition, to treat a particular type of health problem, a physician may use multiple medications, especially in psychiatry. Other factors such as co-morbidities (when more than one condition occurs at the same time, such as ADHD and anxiety) can also influence the number of medications. As a result, the number of drug options can increase rapidly, prolonging the trial-and-error process, as studies on the treatment of major depressive disorder have shown.
- Two out of three patients do not experience relief or remission after the first treatment.[2]
- 38% of patients respond poorly to selective serotonin reuptake inhibitor (SSRI) antidepressants.[3]
- The chance of success decreases with each new treatment trial.[4]
Side effects
Furthermore, certain medications used in treating ADHD can have undesirable side effects that no parent wants their child to suffer:
- Loss of appetite
- Irritability
- Headaches
- Sleep disorders
To minimize these problems, more and more doctors are turning to pharmacogenomics, as it can help them in choosing and dosing a drug.
What is pharmacogenomics?
A combination of pharmacology (study of medications) and genomics (study of the genome), pharmacogenomics studies the influence of genes on the effect of medications. Its aim is to guide the choice of treatment and reduce the trial-and-error process.[5]
Metabolism: Normal, fast or slow
Michel Cameron, Assistant Director of Pharmacogenomics at BiogeniQ, which Biron acquired in 2018, explains that “when a pill is swallowed, it dissolves in the body to produce its effect. It is also metabolized, i.e. transformed by our enzymes (mainly located in the liver) to assist its elimination.”
Dozens of different enzymes are responsible for metabolism, and each has its preferences for one prescribed drug or another. Therefore, the enzymes involved depend on the drug, and their reaction speed can vary from one person to another depending on genetics. For this reason, we make a distinction between normal, fast or slow metabolizers, according to the case. The potential effects of a drug on each type of metabolism are as follows:
- Compared to a normal metabolizer (representing the average person in a population for which standard doses are calculated), a fast metabolizer breaks down a drug more quickly, which may mean that he or she is never exposed to sufficient amounts of the drug to produce the desired therapeutic effect.
- Compared to a normal metabolizer, a slow metabolizer takes longer to metabolize a drug. The drug remains in the body longer and may even accumulate to very high levels, increasing the risk of side effects.
Genetic testing: Leading the way
Using a simple saliva sample (which requires no special preparation), pharmacogenomic testing aims to determine the reaction speeds of the various enzymes responsible for metabolizing drugs. The results can be used to determine whether adjustments to standard doses are warranted or even whether certain drugs should be avoided to limit the risk of complications or inefficacy.[5]
In this video, Michel Cameron explains the value of pharmacogenomics from a therapeutic perspective (French only).
https://www.youtube.com/watch?v=JqAPxXLBBiE
A tool for prescribing the right treatment
Children with ADHD often share similar symptoms, but each child is unique and should receive a treatment adapted to their profile.
Some doctors request pharmacogenomic testing before prescribing medication in order to gather information that will help them personalize the treatment.
These tests provide valuable information about the child’s unique genetic makeup that can be used to:
Assess the risk of adverse effects or ineffectiveness.
ADHD medications fall into two main categories: stimulants and non-stimulants. Although both can be used to treat ADHD, they work differently, and not all children respond in the same way to each. Pharmacogenomics provides insight into how a drug might work depending on the child’s genetics.
Determine what dose of medication to prescribe.
By identifying the individual’s type of metabolism (normal, slow or fast), it is possible to understand how their body breaks down drugs. As a result, adjustments can be made to the dosage to maximize effectiveness and cause the fewest possible side effects.
Genetic markers that tell a story
For about 70% of children diagnosed with ADHD, first-line treatments (psychostimulants based on methylphenidate or amphetamine) quickly succeed in improving concentration, a desired goal for the majority of children. However, 30% are resistant to initial treatments and require multiple trials to find the one that works.[6]
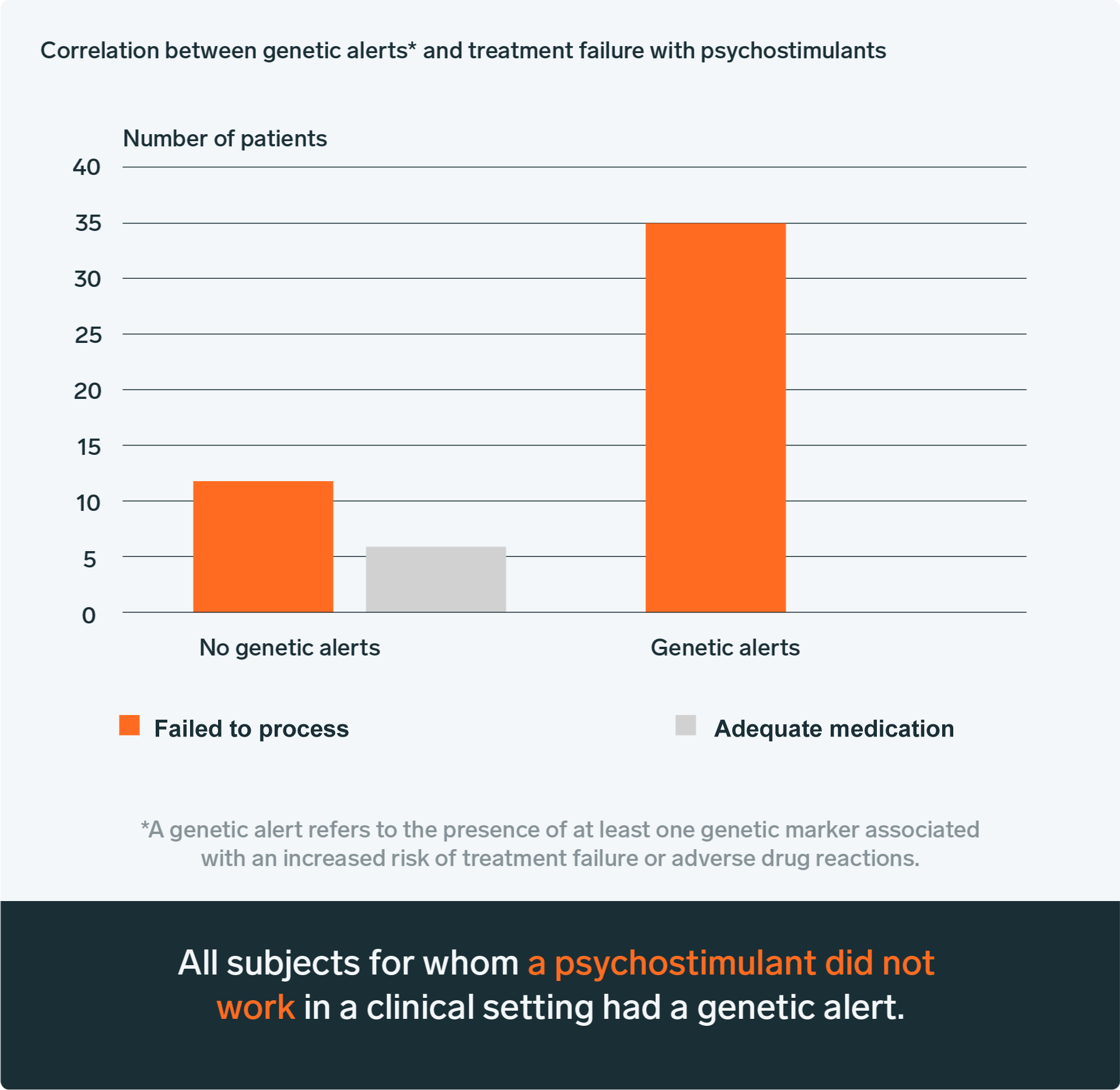
To reduce the number of drug trials, the BiogeniQ laboratory conducted a retrospective study in 2018 on the treatment of ADHD. It demonstrated a link between treatment failures and pharmacogenomic analysis. For example, a pharmacogenomic alert was always present when a treatment had failed in the past, suggesting that the test could be useful in avoiding inappropriate treatments.[7]
A promising science
Although pharmacogenomics is still relatively new, it remains of great interest to practitioners because of the substantial benefits it provides.
Dr. Chad Bousman, a professor of medical genetics at the University of Calgary, leads the PGx-SParK project on the usefulness of pharmacogenomics for children with mental health problems. He believes that a large portion of the trial-and-error process could be avoided thanks to pharmacogenomic testing. His studies have shown that pharmacogenomics increases the chance of remission in cases of major depressive disorders by 41%.[8]
Today, pharmacogenomics provides an additional tool to conventional medicine. It optimizes the trial-and-error process and improves the patient’s condition more quickly, especially in children suffering from ADHD.
BiogeniQ offers pharmacogenomic services that can help your doctor assess the risk of an ADHD medication causing adverse effects or being ineffective.
Sources:
[1] Canadian Paediatric Society. “Medications for attention-deficit hyperactivity disorder,” Caring for kids, May 2019. https://soinsdenosenfants.cps.ca/handouts/behavior-and-development/medications-for-attention-deficit-hyperactivity-disorder
[2] Trivedi, M. H. et al. “Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice,” The American Journal of Psychiatry, Vol. 163, No. 1, p. 28-40, 2006. https://ajp.psychiatryonline.org/doi/full/10.1176/appi.ajp.163.1.28
[3] Spear, B., M. Heath-Chiozzi, J. Huff. “Clinical application of pharmacogenetics,” Trends in Molecular Medicine, Vol. 7, No. 5, p. 201-204, May 2001. http://pkmp.org.pl/assets/31/62/58/66acc37140612d12d81c8ea5b44d3bd33208e4ad.pdf
[4] Huynh, N.N., R. S. McIntyre. “What are the implications of the STAR*D Trial for primary care? A review and synthesis,” Primary Care Companion to the Journal of Clinical Psychiatry, vol. 10, no 2, p. 91-96, 2008. https://www.psychiatrist.com/wp-content/uploads/2021/02/23874_are-implications-stard-trial-primary-care-review-synthesis.pdf
[5] Cameron, M. “Pharmacogenetics for better targeted medication,” Biron Health Group, Feb. 10, 2022. https://www.biron.com/en/education-center/specialist-advice/pharmacogenetics-targeted-medication/
[6] Budur, K. et al. “Non-Stimulant Treatment for Attention Deficit Hyperactivity Disorder,” Psychiatry, Vol. 2, No. 7, p. 4-48, 2005. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3000197/
[7] Cameron, M. “Pharmacogenetics and ADHD: Retrospective study reveals positive results,” Biron Health Group, November 6, 2018. https://www.biron.com/en/news/health-a-to-z/pharmacogenetics-and-adhd-retrospective-study-reveals-positive-results/
[8] Brown, L.C., J.D. Stanton, K. Bharthi, A.A. Maruf, D.J. Müller, C.A. Bousman. “Pharmacogenomic Testing and Depressive Symptom Remission: A Systematic Review and Meta-Analysis of Prospective, Controlled Clinical Trials,” Clinical Pharmacological & Therapeutics, Vol. 112, No. 6, p. 1303-1317, 2022. https://pubmed.ncbi.nlm.nih.gov/36111494/

